Abstract
Introduction: Aggressive chemo-immunotherapy followed by peripheral blood stem cell autografting (ASCT) in CALGB (Alliance) 59909 achieved a median progression-free survival (PFS) in MCL of 5 years (Damon et al JCO, 2009), but late recurrences occurred. Using the CALGB 59909 treatment backbone, we evaluated tolerability and efficacy of adding post-transplant bortezomib consolidation (BC) or maintenance (BM)in a randomized phase II trial. Interim results of CALGB (Alliance) 50403 were previously presented (ASH 2015) demonstrating 5 yr PFS 70% and 69% for the BM and BC arms respectively. We now report final 8 year follow up results from this trial and compare outcomes to CALGB 59909, which differed from CALGB 50403 only by the absence of post-transplant bortezomib.
Methods: The primary endpoint was PFS measured from randomization for each treatment arm. Induction therapy was with 2-3 cycles of augmented R-CHOP (2000 mg/m2 cyclophosphamide) and methotrexate (300 mg/m2) followed by high-dose cytarabine/etoposide/rituximab(R)/filgrastim (EAR) stem cell mobilization and cyclophosphamide/carmustine/etoposide (CBV) ASCT. After 2 doses of post-transplant R, patients were randomized to BC (1.3 mg/ m2 IV days 1, 4, 8, 11 of a 3-week cycle for 4 cycles) or BM (1.6 mg/m2 IV weekly x4 every 8 weeks for 18 months) beginning at approximately day 90. Minimal residual disease (MRD) was analyzed using patient-specific PCR probes for the bcl-1 / IgH junction or the IgH CDR3 region.
Results: 151 patients were enrolled from 10/2006 - 6/2010 at 14 sites; 147 who received treatment are included in this analysis. Median age was 59 years (29-69); stage II (2.7%), III (11.6%), IV (85.0%); MIPI low (52.4%), intermediate (30.6%), high (17.0%); blastoid histology (12.9%); bone marrow involvement (81.0%); Ki67 was evaluated in 86 and was >30% in 17.4% of patients. 118 (80%) underwent ASCT and 102 (68%) were randomized. Most withdrawals (45) were for progression (10) or adverse events (AEs) (19) including 4 treatment-related deaths. Fifty-two patients were randomized to BM and 50 to BC. Following randomization, 34 (65%) completed BM and 33 (66%) completed BC. Withdrawal for AEs occurred in 14 (28%) of BC and 7 (13%) of BM patients (p = 0.09), most for cytopenias or peripheral neuropathy. With a median follow-up 7.8 years from randomization (8.3 from registration), the median PFS from randomization for the BM arm was not reached and was 8.9 years (95% CI 7.2 to not reached) for the BC arm. Both arms had median PFS significantly greater than the null hypothesis setting median PFS to 4 years (p < 0.001; 1-sided test of exponential parameter)The 8-year PFS estimates in the BM and BC arms were 77% (95% CI 66-90%) and 58% (95% CI 44-76%), respectively.
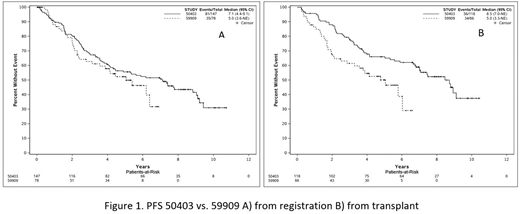
Among all 147 patients treated on CALGB 50403, 8-year PFS from time of registration was 43.6% (35.6-53.3%). PFS from registration was not significantly extended in CALGB 50403 compared with CALGB 59909 (log rank p=0.24), but using a landmark analysis from time of transplant, PFS was significantly extended in CALGB 50403 (log rank p=0.005)(fig 1). Baseline patient characteristics in the two studies were not significantly different. In CALGB 50403, 8-year PFS from registration by MIPI was 52.0% (95% CI 41.1-66.0%) in MIPI low risk, 37.5% (95% CI 25.3-55.4%) in intermediate risk, and 28.2% (95% CI 13.7-57.9%) in high-risk. Bone marrow MRD results were collected for 42 patients post-induction therapy; 8-year PFS estimates were 80.2% (95% CI 62.2-100%) (n=17) and 43.2% (95% CI 27.3-68.2%) (n=25) for MRD-negative and MRD-positive patients, respectively (p=0.009).
Conclusions: Induction chemotherapy followed by ASCT and either BC or BM was efficacious and tolerable, although BC was associated with more withdrawals for toxicity. PFS was not significantly different between BC and BM. The comparison between studies 50403 and 59909 with long-term follow up continues to suggest a PFS benefit from the addition of BC or BM among patients undergoing transplant. This did not translate into a PFS benefit from time of study enrollment possibly due to the higher pre-transplant dropout rate in 50403. MRD-negativity following induction chemo-immunotherapy is highly associated with improved PFS and the role of ASCT in post-induction MRD-negative patients is currently under investigation in a randomized clinical trial.
Support: U10CA180821, U10CA180882, U24CA196171; ClinicalTrials.gov Identifier: NCT00310037
Kaplan:Bayer Pharmaceuticals: Consultancy. Maurer:Celgene: Research Funding; Morphosys: Research Funding; Nanostring: Research Funding. Stock:Jazz Pharmaceuticals: Consultancy. Bartlett:Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees; KITE: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy, Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Blum:Acerta: Consultancy; Astra-Zeneca: Consultancy. LaCasce:Seattle Genetics: Consultancy, Honoraria; Humanigen: Consultancy, Honoraria; Research to Practice: Speakers Bureau; Bristol-Myers Squibb: Other: Data safety and monitoring board. Leonard:AstraZeneca: Consultancy; Juno: Consultancy; BMS: Consultancy; ADC Therapeutics: Consultancy; Gilead: Consultancy; Biotest: Consultancy; Karyopharm: Consultancy; Novartis: Consultancy; MEI Pharma: Consultancy; Celgene: Consultancy; United Therapeutics: Consultancy; Sutro: Consultancy; Genentech/Roche: Consultancy; Pfizer: Consultancy; Bayer: Consultancy. Cheson:AbbVie, Roche/Genentech, Pharmacyclics, Acerta, TG Therapeutics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal